Definition of lipid
Lipid are compound that occur frequently. The definition of lipid is base on solubility. Lipids are marginally soluble (at best) in water but readily soluble in organic solvent, such as chloroform or acetone. In terms of chemistry, lipids are a mixed of bag of compounds that share some properties based on structural similarities, mainly preponderance of nonpolar groups.
2 main groups of lipid:
FATTY ACID
Lipid are compound that occur frequently. The definition of lipid is base on solubility. Lipids are marginally soluble (at best) in water but readily soluble in organic solvent, such as chloroform or acetone. In terms of chemistry, lipids are a mixed of bag of compounds that share some properties based on structural similarities, mainly preponderance of nonpolar groups.
2 main groups of lipid:
- one group which consist of open-chain compounds with polar head groups and long non-polar tail includes fatty acids,triacylglycrol, sphingolipids, phosphoacylglycerol, glycolipid
- second major group consist of fused ring compounds, the steroids; an important representative of this group is cholestrol.
FATTY ACID
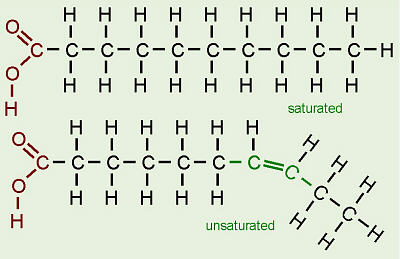
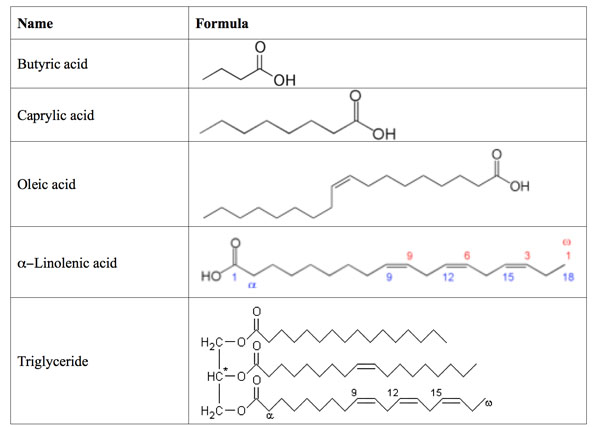
- A fatty acids has a carrboxyl group at the polar end and a hydrocarbon chain at the nonpolar tail.
- amphipatic ( carboxyl group is hydrophilic and the hydrocarbon til is hydrophobic.
- if there are C=C double bond in the chain, the fatty acid is unsaturated, if there are C-C single bond, the fatty acid is saturated
TRIACYLGLYCEROLS
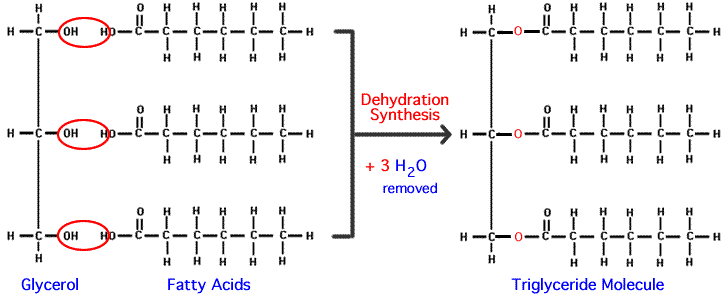
- Simple compund that contains three hydroxyl groups
- when all the three alcohol group form ester linkage with fatty acid, the resulting compound called triacylglycerol (triglyceride)
- three ester groups are polar part
- tail of fatty acid are non polar
- triacylglycerols accumulate in adiose tissue and provide a means of storing fatty acids, particularly in animals.
- when organism uses fatty acids, the ester linkages of triacylglycerols are hydrolyzed by enzymes called lipases.
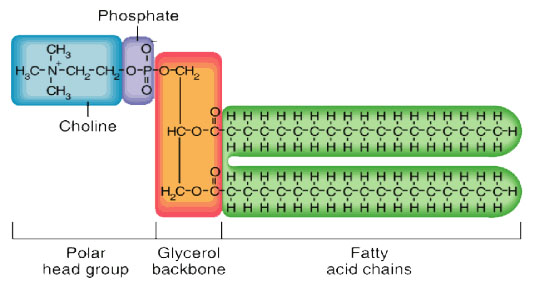
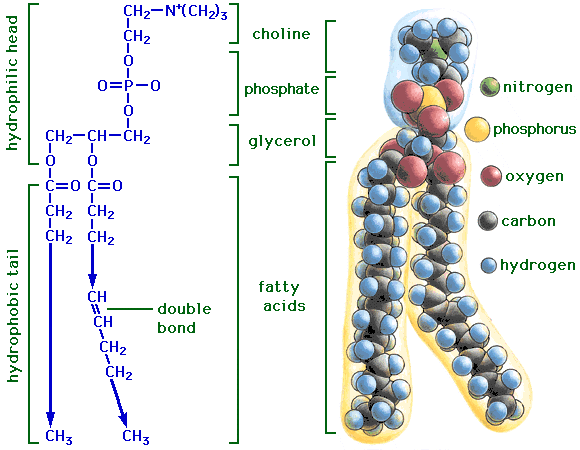
Phosphoacylglycerols (phospholipids)
- It is possible for one of the alcohol groups of glycerol to be esterified by a phosphoric acid molecule rather than by a carboxylic acid. In such lipid molecule, two fatty acids are aso esterified to the glycerol molecule. The resulting compound is called a phosphatidic acid.
- fatty acids are usually monoprotic acid with only one carboxyl group able to form an ester bond, but phosphoric acid is triprotic and thus can form more than one ester linkage.
- one molecule of phosphoric acid can form ester bond both to glycerol and some other alcohol, creating s phosphatidyl ester. Phosphatidyl ester are classed as phosphoacylglcerols.
- the classification of a phosphatidyl ester depends on the nature of the second alcohol esterified to the phosphoric acid.
- some of the important lipids in this class are: cephalin, phosphatidyl serine, lecithin, phosphatidyl ethanolamine, phosphatidyl glycerol and cardiolipin.
WAXES
GLYCOLIPIDS
- complex mixture of esters a long-chain carboxylic acids and long chain alcohol
- serve as protective coating for both plants and animals.
GLYCOLIPIDS
- When carbohydrate is bound to an alcohol group of a lipid by a glycosidic linkage, the resulting coumpound is glycolipid.
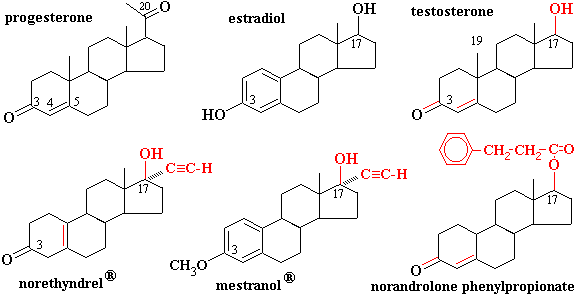
STEROIDS
- Many compounds of widely differing functions are steroids because they have the same general structure: a fused-ring system consisting of three six-membered rings and one five-membered ring
Mary K. Campbell, Shawn O. Farrel. Biochemistry: fifth edition. THOMSON, United States of America. 2006
lipid metabolism
- Lipolysis, the removal of the fatty acid chains from the glycerol to which they are bound in their storage form as triglycerides (or fats), is carried out by lipases.
- Once freed from glycerol, the free fatty acids enter the blood, which transports them, attached to plasma albumin, throughout the body.
- Long chain free fatty acids enter the metabolizing cells (i.e. most living cells in the body except red blood cells and neurons in the central nervous system) through specific transport proteins, such as the SLC27 family fatty acid transport protein. Red blood cells do not contain mitochondria and are therefore incapable of metabolizing fatty acids; the tissues of the central nervous system cannot use fatty acids, despite containing mitochondria, because fatty acids cannot cross the blood brain barrier into the interstitial fluids that bathe these cells.
- Once inside the cell long-chain-fatty-acid—CoA ligase catalyzes the reaction between a fatty acid molecule with ATP (which is broken down to AMP and inorganic pyrophosphate) to give a fatty acyl-adenylate, which then reacts with free coenzyme A to give a fatty acyl-CoA molecule.
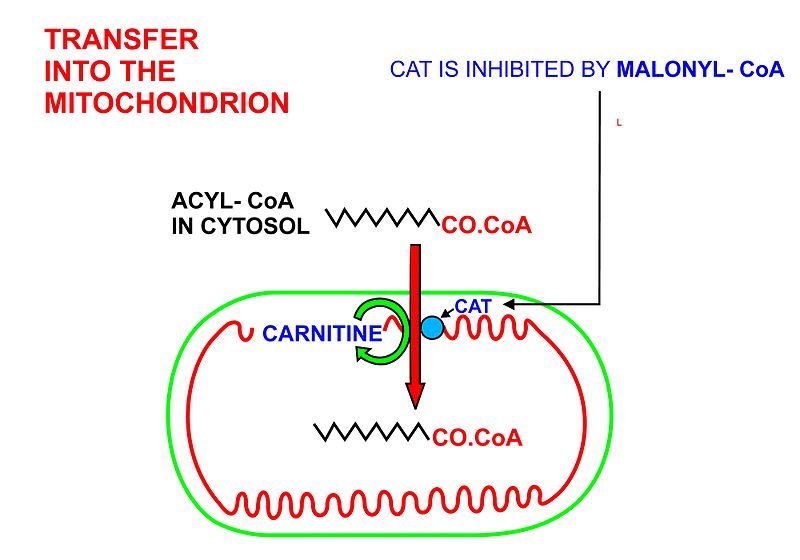
- In order for the acyl-CoA to enter the mitochondrion the carnitine shuttle is used:
- Acyl-CoA is transferred to the hydroxyl group of carnitine by carnitine palmitoyltransferase I, located on the cytosolic faces of theouter and inner mitochondrial membranes.
- Acyl-carnitine is shuttled inside by a carnitine-acylcarnitine translocase, as a carnitine is shuttled outside.
- Acyl-carnitine is converted back to acyl-CoA by carnitine palmitoyltransferase II, located on the interior face of the inner mitochondrial membrane. The liberated carnitine is shuttled back to the cytosol, as an acyl-CoA is shuttled into the matrix.
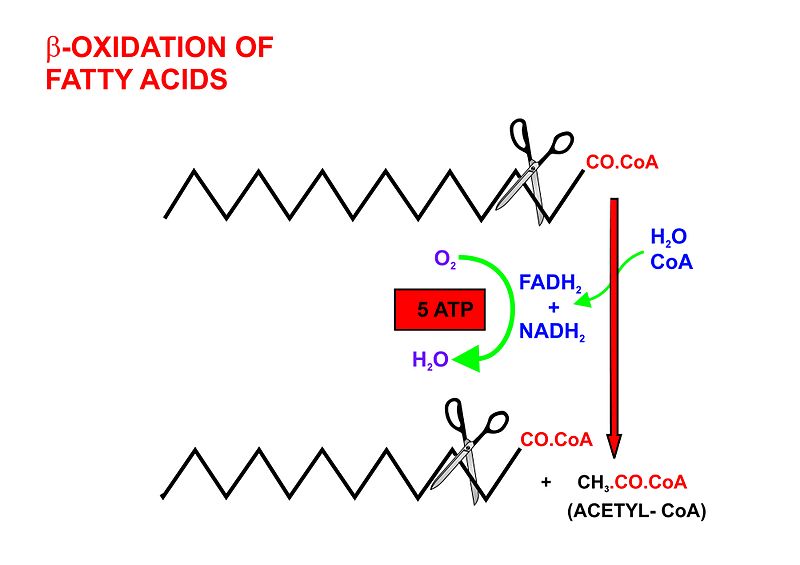
- Beta oxidation, in the mitochondrial matrix, then cuts the long carbon chains of the fatty acids (in the form of acyl-CoA molecules) into a series of two-carbon (acetate) units, which, combined with co-enzyme A, form molecules of acetyl CoA, which condense withoxaloactetate to form citrate at the "beginning" of the citric acid cycle. It is convenient to think of this reaction as marking the "starting point" of the cycle, as this is when fuel - acetyl-CoA - is added to the cycle, which will be dissipated as CO2 and H2O with the release of a substantial quantity of energy captured in the form of ATP, during the course of each turn of the cycle.
- Dehydrogenation by acyl-CoA dehydrogenase, yielding 1 FADH2
- Hydration by enoyl-CoA hydratase
- Dehydrogenation by 3-hydroxyacyl-CoA dehydrogenase, yielding 1 NADH + H+
- Cleavage by thiolase, yielding 1 acetyl-CoA and a fatty acid that has now been shortened by 2 carbons (forming a new, shortened acyl-CoA)
- The acetyl-CoA produced by β-oxidation enters the citric acid cycle in the mitochondrion by combining with oxaloacetate to form citrate. This results in the complete combustion of the acetyl-CoA to CO2 and water. The energy released in this process is captured in the form of 1 GTP and 11 ATP molecules per acetyl-CoA molecule oxidized.[1][7] This is the fate of acetyl-CoA wherever β-oxidation of fatty acids occurs, except under certain circumstances in the liver. In the liver oxaloacetate can be wholly or partially diverted into thegluconeogenic pathway during fasting, starvation, a low carbohydrate diet, prolonged strenuous exercise, and in uncontrolled type 1 diabetes mellitus. Under these circumstances oxaloacetate is hydrogenated to malate which is then removed from the mitochondrion to be converted into glucose in the cytoplasm of the liver cells, from where it is released into the blood.In the liver, therefore, oxaloacetate is unavailable for condensation with acetyl-CoA when significant gluconeogenesis has been stimulated by low (or absent) insulin and high glucagon concentrations in the blood. Under these circumstances acetyl-CoA is diverted to the formation of acetoacetate and beta-hydroxybutyrate.Acetoacetate, beta-hydroxybutyrate, and their spontaneous breakdown product, acetone, are frequently, but confusingly, known as ketone bodies (as they are not "bodies" at all, but water-soluble chemical substances). The ketone bodies are released by the liver into the blood. All cells with mitochondria can take ketone bodies up from the blood and reconvert them into acetyl-CoA, which can then be used as fuel in their citric acid cycles, as no other tissue can divert its oxaloacetate into the gluconeogenic pathway in the way that this can occur in the liver. Unlike free fatty acids, ketone bodies can cross the blood-brain barrier and are therefore available as fuel for the cells of the central nervous system, acting as a substitute for glucose, on which these cells normally survive. The occurrence of high levels of ketone bodies in the blood during starvation, a low carbohydrate diet, prolonged heavy exercise and uncontrolled type 1 diabetes mellitus is known as ketosis, and, in its extreme form, in out-of-control type 1 diabetes mellitus, as ketoacidosis.