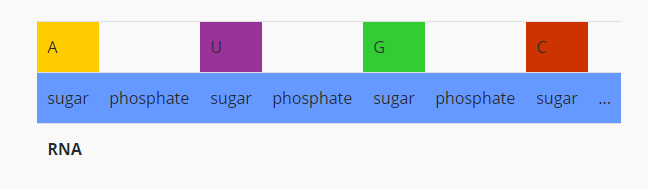
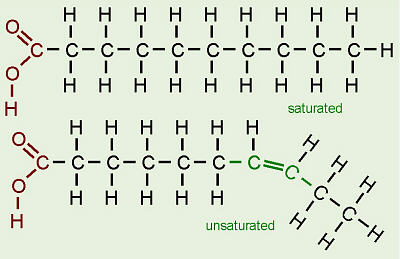
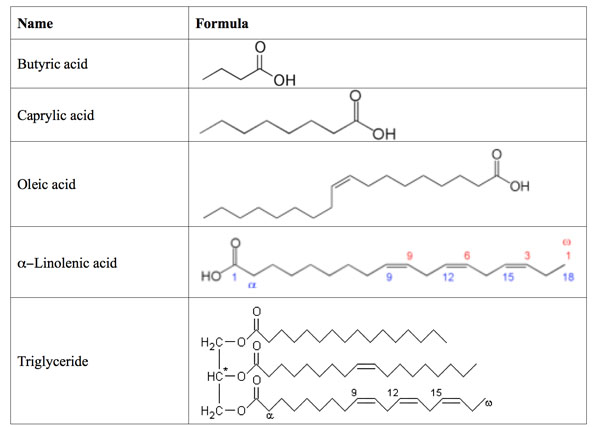
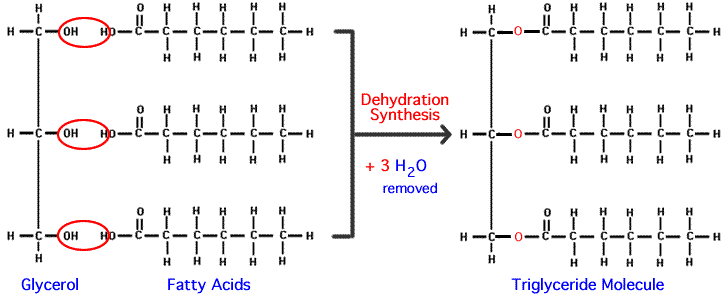
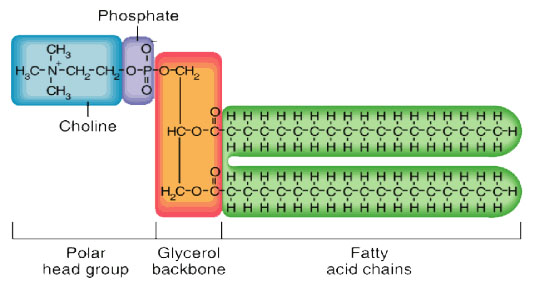
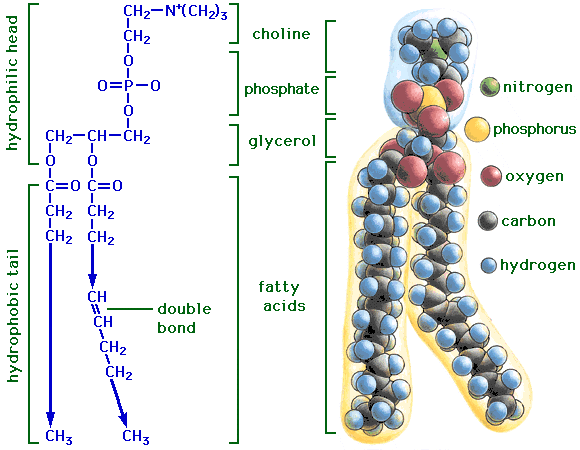
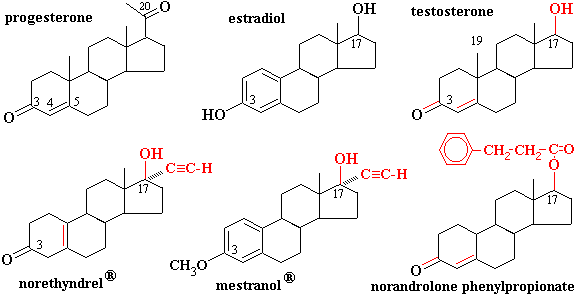
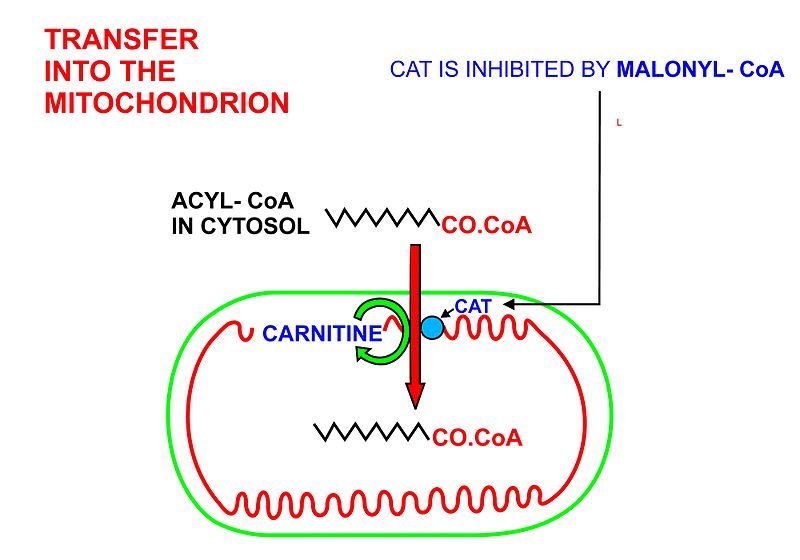
How does the structure of protein determine their function?
Ex: primary structure, secondary structure, tertiary structure, quaternary structure
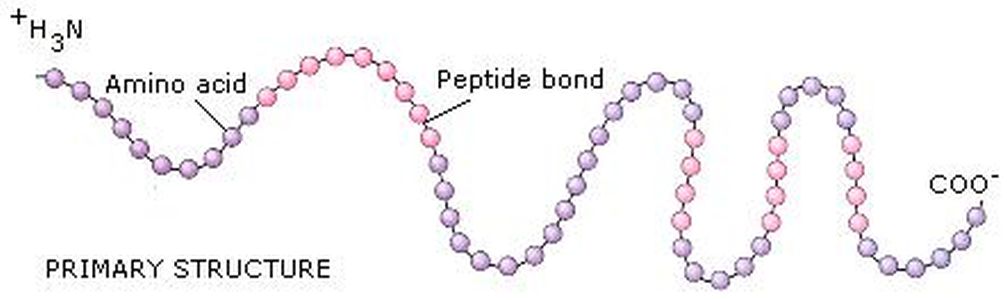
Primary structure is the order in which the amino acids are covalently llinked together. The peptide Leu-Gly-Thr-Val-Arg-Asp-His has a different primary structure from the peptide Val-His-Asp-Leu-Gly-Arg-Thr, even though both have the same number and kinds of amno acids. Primary structure is one dimensional first step in specifying the three-dimensional structure of protein.
Two three-dimensional aspects of a single polypeptide chain, called the secondary and tertiary structure, can be considered separately. Secondary structure is the arrangement in space of the atoms in the peptide backbone. The α-helix and β-pleated sheet arrangement are two different types of secondary structure. Secondary structures have repetitive interactions resulting from hydrogen bonding between the amide N-H and the carbonyl groups of the peptide backbone
Tertiary structure include three-dimensional arrangement of all the atoms in the protein, including those in the side chains and in any prosthetic groups (groups of atoms other than amino acids)
Tertiary structure includes the three-dimensional arrangement chains called subunits. The arrangement of subunits with respect to one another is the quaternary structure. Interaction between subunits is mediated by noncovalent interactions, such as hydrogen bonds, electrostatic interactions, and hydrophobic interactions.
PRIMARY STRUCTURE
- Biologically active proteins are polymers consisting of amino acids linked by covalent peptide bonds.
- Many proteins have no obvious regular repeating structure. As a sequence, these proteins are frequently described as having large segments of "random structure"(also referred to as random coil).
- Proteins are complex, they are defined in terms of four levels of structure.
Ex: primary structure, secondary structure, tertiary structure, quaternary structure
Primary structure is the order in which the amino acids are covalently llinked together. The peptide Leu-Gly-Thr-Val-Arg-Asp-His has a different primary structure from the peptide Val-His-Asp-Leu-Gly-Arg-Thr, even though both have the same number and kinds of amno acids. Primary structure is one dimensional first step in specifying the three-dimensional structure of protein.
Two three-dimensional aspects of a single polypeptide chain, called the secondary and tertiary structure, can be considered separately. Secondary structure is the arrangement in space of the atoms in the peptide backbone. The α-helix and β-pleated sheet arrangement are two different types of secondary structure. Secondary structures have repetitive interactions resulting from hydrogen bonding between the amide N-H and the carbonyl groups of the peptide backbone
Tertiary structure include three-dimensional arrangement of all the atoms in the protein, including those in the side chains and in any prosthetic groups (groups of atoms other than amino acids)
Tertiary structure includes the three-dimensional arrangement chains called subunits. The arrangement of subunits with respect to one another is the quaternary structure. Interaction between subunits is mediated by noncovalent interactions, such as hydrogen bonds, electrostatic interactions, and hydrophobic interactions.
PRIMARY STRUCTURE
- The amino acids sequence (primary structure) of a protein determines its three-dimensional structure, which, in turn, determines its properties.
- One of the most striking demonstrations of the importance of primary structure is found in the hemoglobin associated with sickle-cell anemia. In this genetic disease, red blood cells cannot bind oxygen efficiently.
SECONDARY STRUCTURE
- The secondary structure of proteins is the hydrogen-bonded arrangement of the backbone of the protien, the polypeptide chain
- The α-helix and β-pleated sheet are periodic structures; their features repeat at regular intervals. The α-helix is rodlike and involves only one one polypeptide chain. The β-pleated sheet structure can give a two-dimensional array and can involve one or more polypeptide chains.
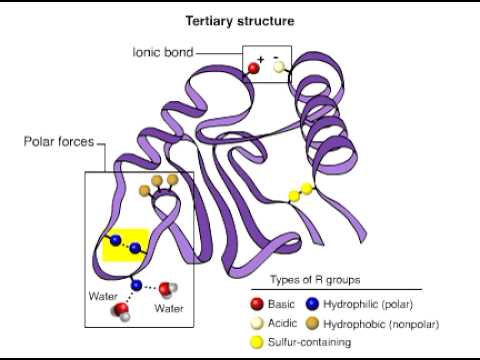
TERTIARY STRUCTURE
- The tertiary structure will have a single polypeptide chain "backbone" with one or more protein secondary structure, the protein domain. Amino acid side chains may interact and bond in a number of ways. The interactions and bonds of side chains within a particular protein determine its tertiary structure. The protein tertiary structure is defined by its atomic coordinates. These coordinates may refer either to a protein domain or to the entire tertiary structure. A number of tertiary structures may fold into a quaternary structure
- Globulr proteins have a core of hydrophobic amino acid residues and a surface region of water-exposed, charged, hydrophilic residues. This arrangement may stabilise interactions within the tertiary structure
QUATERNARY STRUCTURE
- Quaternary Structure is the combination of two or more chains, to form a complete unit. The interactions between the chains are not different from those in tertiary structure, but are distinquished only by being interchain rather than intrachain.
- Some proteins are composed of identical subunits (chains). A simple example is the dimer of HIV Protease
- Some proteins are composed of non-identical subunits (chains). A simple example is insulin which is made up of two chains, the alpha chain and the beta chain, linked by two disulfide bridges.
- Even with identical subunits, their relative positioning controls the symmetry of the complex and can have biochemical implications. Consider two right hands, nearly identical to each other but completely assymetric.
Mary K. Campbell, Shawn O. Farrel. Biochemistry: fifth edition. THOMSON, United States of America. 2006
biochemistry of protein
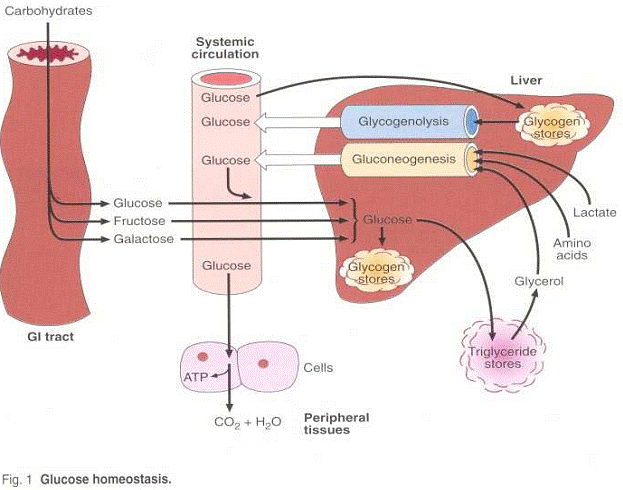
During protein metabolism, some protein is converted to glucose in a process called gluconeogenesis, the formation of glucose from non-carbohydrate sources
The basic difference between protein and carbohydrate is that while carbohydrates are made out of simple sugars (carbon, hydrogen, and oxygen), protein is made from amino acids (carbon, hydrogen, oxygen, nitrogen, and sufur). The nitrogen is a basic component of the protein's amino acids and accounts for 13 to 20% of the total mass.
The first step in protein metabolism is to break it into its constituent amino acids. These are absorbed into the blood stream.
The second step is to break down the amino acids into their constituent parts--catabolism, if you want to get technical about it. This removes the nitrogen or amino group from the amino acids. The process is called deamination.
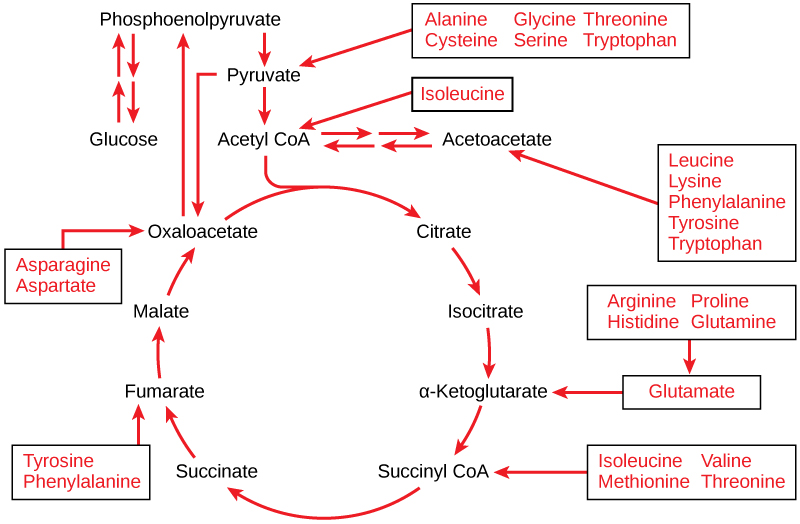
Deamination breaks the amino group down into ammonia and what is termed the carbon skeleton. Ammonia is converted to urea, filtered through the kidneys, and excreted in urine. The carbon skeleton--which is composed of carbon, hydrogen, and oxygen--can then by used either for protein synthesis, energy production (ATP), or converted to glucose by gluconeogenesis.
Most authorities believe that the amount of protein converted to glucose is quite small, except under conditions of intense exercise or metablic starvation. Under these conditions amino acids produce the major source of glucose for blood sugar maintenance.
The basic difference between protein and carbohydrate is that while carbohydrates are made out of simple sugars (carbon, hydrogen, and oxygen), protein is made from amino acids (carbon, hydrogen, oxygen, nitrogen, and sufur). The nitrogen is a basic component of the protein's amino acids and accounts for 13 to 20% of the total mass.
The first step in protein metabolism is to break it into its constituent amino acids. These are absorbed into the blood stream.
The second step is to break down the amino acids into their constituent parts--catabolism, if you want to get technical about it. This removes the nitrogen or amino group from the amino acids. The process is called deamination.
Deamination breaks the amino group down into ammonia and what is termed the carbon skeleton. Ammonia is converted to urea, filtered through the kidneys, and excreted in urine. The carbon skeleton--which is composed of carbon, hydrogen, and oxygen--can then by used either for protein synthesis, energy production (ATP), or converted to glucose by gluconeogenesis.
Most authorities believe that the amount of protein converted to glucose is quite small, except under conditions of intense exercise or metablic starvation. Under these conditions amino acids produce the major source of glucose for blood sugar maintenance.
In a person without diabetes, a rise in blood amino acid concentration (the result of protein metabolism) stimulates the secretion of both glucagon and insulin, so their blood sugar remains stable. But in people with diabetes, the release of glucagon without insulin or with impaired insulin response can cause our blood sugar to rise precipitously several hours after a meal high in protein.
The insulin is secreted to stimulate protein synthesis--the uptake of amino acids into muscle cells--making them less available for gluconeogenesis. The glucagon is secreted to stimulate the uptake of amino acids into the cells of the liver for gluconeogenesis.
So why are these two hormones battling for opposing uses of the same amino acids? Isn't that non-productive?
Actually, the phenomenon serves an important purpose. As you probably know, insulin lowers the blood sugar, while glucagon raises it. In the non-diabetic state, the release of these two opposing hormones ensures that the amino acids are used for protein synthesis (because of the extra insulin) but the blood sugar doesn't drop to dangerously low levels, even if the meal was low in carbohydrate. As a result, blood glucose concentration remains reasonably stable during protein metabolism. The insulin and glucagon essentially cancel each other out in terms of their effect on blood glucose, while the insulin is still able to promote protein synthesis.
But in people with diabetes, as I mentioned earlier, the release of glucagon without insulin or with impaired insulin response can cause our blood sugar to rise precipitously several hours after a meal high in protein. This is due not only to the glucagon's directly raising the blood sugar, but also to the fact that in the absence of insulin it increases the amount of the amino acids that are used for gluconeogenesis.
For what it's worth, arginine is the amino acid reputed to stimulate the highest production of both insulin and glucagon.
The insulin is secreted to stimulate protein synthesis--the uptake of amino acids into muscle cells--making them less available for gluconeogenesis. The glucagon is secreted to stimulate the uptake of amino acids into the cells of the liver for gluconeogenesis.
So why are these two hormones battling for opposing uses of the same amino acids? Isn't that non-productive?
Actually, the phenomenon serves an important purpose. As you probably know, insulin lowers the blood sugar, while glucagon raises it. In the non-diabetic state, the release of these two opposing hormones ensures that the amino acids are used for protein synthesis (because of the extra insulin) but the blood sugar doesn't drop to dangerously low levels, even if the meal was low in carbohydrate. As a result, blood glucose concentration remains reasonably stable during protein metabolism. The insulin and glucagon essentially cancel each other out in terms of their effect on blood glucose, while the insulin is still able to promote protein synthesis.
But in people with diabetes, as I mentioned earlier, the release of glucagon without insulin or with impaired insulin response can cause our blood sugar to rise precipitously several hours after a meal high in protein. This is due not only to the glucagon's directly raising the blood sugar, but also to the fact that in the absence of insulin it increases the amount of the amino acids that are used for gluconeogenesis.
For what it's worth, arginine is the amino acid reputed to stimulate the highest production of both insulin and glucagon.