Learning outcomes:
1. water structural molecule
2. hydorgen bond in water molecule
3. thermal and solvent properties in water
4. ionzation of water
Water is a simple molecule with two hydrogen atoms and an oxygen atom. Its molecular weight is 18. Water has unique features that cause it to behave differently than other compounds of similar molecular weight. The water molecule is dipolar because it has a negatively charged side and a positively charges one. Polarity leads to water having a high freezing and boiling points, large latent heat requirement for phase changes between ice and liquid and between liquid and vapour, temperature dependant density, a large capacity to hold heat, and good solvent action. The physical properties of water influence water quality.
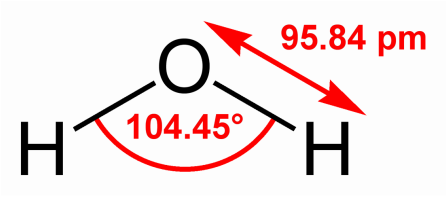
A water molecule consists of two hydrogen atoms covalently bonded to one oxygen atom. The angle formed by lines through the centers of hydrogen nuclei and oxygen nucleus is 104.45 degree. An oxygen nucleus is heavier than a hydrogen nucleus, so electrons are pulled relatively closer to the oxygen nucleus. This gives the oxygen atom a small negatively charges and each of the hydrogen atoms a slight positive charge resulting in polarity of the water molecule.
Molecules attract each other through van der waals forces. Negatively charged electrons of one molecule and positively charged nuclei of another molecule attract. This attraction is almost but not completely, neutralized by repulsion of electrons by electrons and nuclei by nuclei, and van der wals forces are weak.
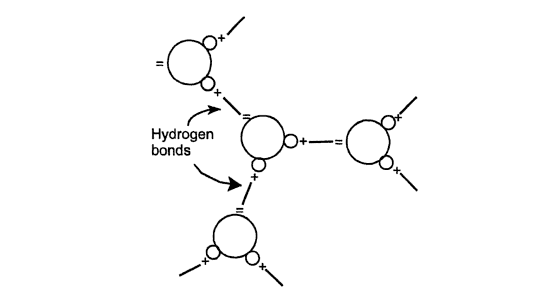
The positively charged side of a water molecule attracts the negatively charged side of another to form dimensions, but hydrogen bonding actually is illustrated here in two dimensions, but hydrogen bonding actually is three-dimensional. One water molecule can be bonded to another with the axis of attraction extending in any direction. Hydrogen bonds are not as strong as covalent bonds or ionic bonds, but they are much stronger than van der waals attraction. The degree of molecular attraction in water is much greater than in nonpolar substances.
1. water structural molecule
2. hydorgen bond in water molecule
3. thermal and solvent properties in water
4. ionzation of water
Water is a simple molecule with two hydrogen atoms and an oxygen atom. Its molecular weight is 18. Water has unique features that cause it to behave differently than other compounds of similar molecular weight. The water molecule is dipolar because it has a negatively charged side and a positively charges one. Polarity leads to water having a high freezing and boiling points, large latent heat requirement for phase changes between ice and liquid and between liquid and vapour, temperature dependant density, a large capacity to hold heat, and good solvent action. The physical properties of water influence water quality.
A water molecule consists of two hydrogen atoms covalently bonded to one oxygen atom. The angle formed by lines through the centers of hydrogen nuclei and oxygen nucleus is 104.45 degree. An oxygen nucleus is heavier than a hydrogen nucleus, so electrons are pulled relatively closer to the oxygen nucleus. This gives the oxygen atom a small negatively charges and each of the hydrogen atoms a slight positive charge resulting in polarity of the water molecule.
Molecules attract each other through van der waals forces. Negatively charged electrons of one molecule and positively charged nuclei of another molecule attract. This attraction is almost but not completely, neutralized by repulsion of electrons by electrons and nuclei by nuclei, and van der wals forces are weak.
The positively charged side of a water molecule attracts the negatively charged side of another to form dimensions, but hydrogen bonding actually is illustrated here in two dimensions, but hydrogen bonding actually is three-dimensional. One water molecule can be bonded to another with the axis of attraction extending in any direction. Hydrogen bonds are not as strong as covalent bonds or ionic bonds, but they are much stronger than van der waals attraction. The degree of molecular attraction in water is much greater than in nonpolar substances.
Water is a liquid between 0 to 100 degree Celsius at standard atmospheric pressure (760 mm of mercury). Freezing and boiling points of water, 0 and 100 degree Celsius respectively, are much higher than those of other hydrogen compounds of low molecular weight, methane, ammonia, phosgene, and hydrogen sulphide, that are gases at ordinary temperatures on the earth’s surface. Molecules of other common hydrogen compounds do not form hydrogen bonds and joined only by Vander Waals attractions. Considerable thermal energy is required to break hydrogen bonds and convert ice to liquid water or to change liquid water to vapour.
Depending its internal content, water exist in solid, liquid, or gaseous phase. In ice, all hydrogen atoms are bonded; in liquid phase, a portion of the hydrogen atoms is bonded; in vapour, there are no hydrogen bonds. An increase in the internal energy content of water agitates its molecules, causes hydrogen bonds to stretch and break, and temperature to rise. The opposite effect occurs when energy content of water declines.
Water freezes when its energy content declines and molecular motion slows so that hydrogen bonds form to produce ice. Ice melts when its energy content rises and molecular motion increases and too few hydrogen bonds are present to maintain the crystalline structure of ice. The freezing point of water is 0 degree Celsius.
Water changes from liquid to vapour when it attains enough internal energy and molecular motion to break all hydrogen bonds. Water vapour condenses to form liquid water when it loses energy and molecular motion decreases to permit formation of hydrogen bonds.
Depending its internal content, water exist in solid, liquid, or gaseous phase. In ice, all hydrogen atoms are bonded; in liquid phase, a portion of the hydrogen atoms is bonded; in vapour, there are no hydrogen bonds. An increase in the internal energy content of water agitates its molecules, causes hydrogen bonds to stretch and break, and temperature to rise. The opposite effect occurs when energy content of water declines.
Water freezes when its energy content declines and molecular motion slows so that hydrogen bonds form to produce ice. Ice melts when its energy content rises and molecular motion increases and too few hydrogen bonds are present to maintain the crystalline structure of ice. The freezing point of water is 0 degree Celsius.
Water changes from liquid to vapour when it attains enough internal energy and molecular motion to break all hydrogen bonds. Water vapour condenses to form liquid water when it loses energy and molecular motion decreases to permit formation of hydrogen bonds.
hydrogen bonds between water molecule
Solvent Action
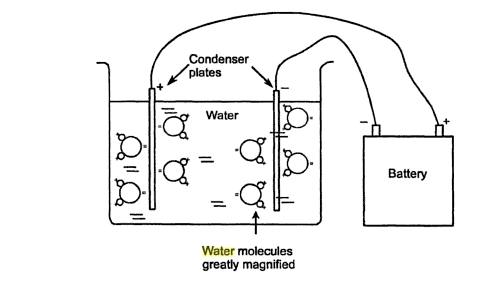
In electric field of a condenser, water molecules orient themselves, the pointing their positive ends toward the negative plate and their negative ends toward the positive plate. The orientation of molecules neutralized part of the charge applied to the condenser plates. The voltage required to produce a given voltage on condenser plates is a measure of the dielectric constant of the substance surrounding the condenser plates. In a vacuum, 1 volt applied from a battery to the plates produces a voltage of 1 volt on the condenser plates. In contrast, the dielectric constant f air and water are 1.0006 volt and 81 volt, respectively.
In electric field of a condenser, water molecules orient themselves, the pointing their positive ends toward the negative plate and their negative ends toward the positive plate. The orientation of molecules neutralized part of the charge applied to the condenser plates. The voltage required to produce a given voltage on condenser plates is a measure of the dielectric constant of the substance surrounding the condenser plates. In a vacuum, 1 volt applied from a battery to the plates produces a voltage of 1 volt on the condenser plates. In contrast, the dielectric constant f air and water are 1.0006 volt and 81 volt, respectively.
Claude E.boyd. An Introduction: Water Quality. USA: Kluwer Academic Publisher. (2000)