Learning outcome:
1. oxidation and reduction
2. Role of ATP
3. Law of Thermodynamic
REDOX
The half-reactions must be combined in a way that preserves electrical neutrality. There can be no net gain or loss of electrons. In determining the direction of an overall reaction, keep in mind the sign convention for reduction potentials. According to this convention, a half-reaction having a highly negative reduction potential involves a strong reducing agent; one having a highly positive reduction potential involves a strong oxidizing agent:
1. oxidation and reduction
2. Role of ATP
3. Law of Thermodynamic
REDOX
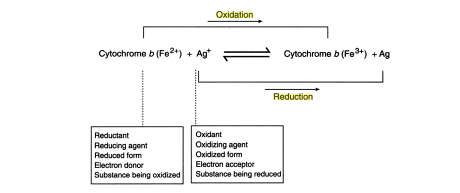
- Oxidation-reduction (redox) reactions involve changes in the electronic structure of atoms and molecules.
- Oxidation represents a loss of electrons (e-), and reduction a gain of electrons.
- Electron losses and gains result from direct transfer of electron from one substance to another or from transfer of electrons in association with an H, an O, or some other atom.
- Because hydrogen generally acts as an electropositive element and oxygen as an electronegative element, gain of oxygen and loss of hydrogen are equivalent to oxidation.
The half-reactions must be combined in a way that preserves electrical neutrality. There can be no net gain or loss of electrons. In determining the direction of an overall reaction, keep in mind the sign convention for reduction potentials. According to this convention, a half-reaction having a highly negative reduction potential involves a strong reducing agent; one having a highly positive reduction potential involves a strong oxidizing agent:
Of two half-reactions, the one having the smaller reduction potential involves a stronger reducing agent than the other half-reaction. Consequently, the half reaction having the smaller reduction potential proceeds as an oxidation (loss of electrons) and must be written in the reverse sense. The electrons generated by this half-reaction cause the second half-reaction to proceed as a reduction (gain electron). These relationships hold regardless of the signs of potentials. Thus, of two half-reactions having reduction potentials of -0.52V and -0.27V, respectively, the former will proceed as and oxidation, the latter as a reduction. Likewise, of two half-reactions having reduction potentials of +0.18V and +0.46V, respectively, the former will proceed as an oxidation, the latter as a reduction. With this in mind, let us proceed to couple two half-reactions, once under standard and once under actual conditions.
J.Stenesh. Biochemistry (4th Edition). 1998. United States in America: Plenum Press.
ROLE OF ATP
Most of what a cell does involves endergonic reactions, requiring constant energy input; these reactions are coupled to just a few exergonic reactions which supply that energy.
Most of what a cell does involves endergonic reactions, requiring constant energy input; these reactions are coupled to just a few exergonic reactions which supply that energy.
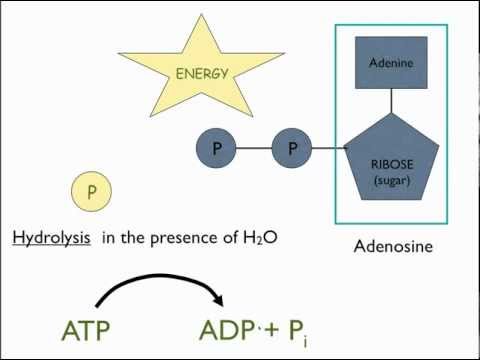
- The key fuel source in cell reactions is Adenosine Triphosphate, or ATP.
- The molecule is an amino acid linked to a ribose sugar, linked to a string of three phosphate groups (there are also ADP, Adenosine Diphosphate, and AMP, Adenosine Monophosphate).
- With ATP, the breaking off of the last phosphate, often in a transfer to another molecule (a process called phosphorylation), can release or activate a fair amount of usable energy.
- A couple of other molecules are important as electron-carrying molecules. Nicotinamide Adenine Dinucleotide, or NAD, a coenzyme derived from the vitamin niacin, or B3, is important in reduction and oxidation (often called redox) reactions. When an atom or molecule is oxidized, it loses electrons; when one is reduced, it gains electrons (whose negative charges would reduce the charge of the recipient).
- In the case of NAD, its NAD+ form picks up H2 (by taking it from another source, it is an oxidizing agent) and briefly becomes NADH2+, then quickly stabilizes by losing H+ and becoming NADH. This molecule can return to NAD+ by losing a hydrogen and an electron (it is called a reducing agent in this role), allowing the electron to move elsewhere. Then, with phosphorylation to NADPH, another important molecule, especially in the process of photosynthesis, is made.
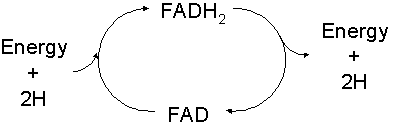
- Flavin-Adenine Dinucleotide, or FAD, another coenzyme, this one derived from riboflavin / B2, serves a similar function. FAD can be reduced to FADH2 and carry hydrogens / electrons between reactions. The main sources of ATP in most cells are fueled by hydrogens carrying electrons.
retrieved from http://faculty.fmcc.suny.edu/mcdarby/majors101book/Chapter_04-Cell_Structure&Function/04-cells&energy.htm
|
LAW OF THERMODYNAMIC
• Thermochemistry is a branch of thermodynamics • Thermodynamics – takes into account all energy effects • Two types of energies involved: Heat (q) and Work (w). First Law of thermodynamics - conservation of energy. Energy can not be created or destroyed, it is transferable |
DE system = - DE surroundings
DE = q + w |
|
Nature of energy
• Work (w) • Make objects with mass to move a distance against a force |
W = Force x distance
= m x g x d W = Pressure x volume |
|
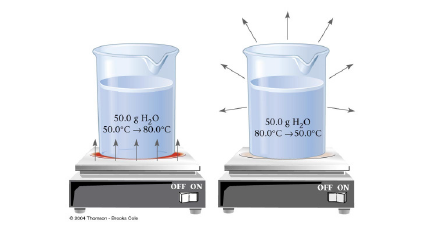
• Heat (q)
• Energy used to cause temperature to increase |
q = m x s x sum of T
|
Principles of Heat Flow
• System – Chemical
• Surroundings – everything else
• System – Chemical
• Surroundings – everything else
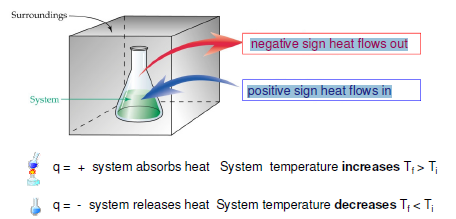
Direction and sign of heat flow
Heat is given symbol “q”
Heat is given symbol “q”
Magnitude of heat flow
• Units for heat flow
1 cal = 4.184 J
This is the amount of heat required to raise the temperature of one gram of water by one degree Celsius
• Units for heat flow
1 cal = 4.184 J
This is the amount of heat required to raise the temperature of one gram of water by one degree Celsius
Note: nutritionists use the term “Calorie” = kcal
2000 Calorie diet day helps lose a few pounds.
(2000 kcal = 200,000 cal of energy)
Calculating heat flow (q)
• Heat flow – changing the temp of system
q = C x DT
heat = (heat capacity) x (Tfinal – Tinitial)
(2000 kcal = 200,000 cal of energy)
Calculating heat flow (q)
• Heat flow – changing the temp of system
q = C x DT
heat = (heat capacity) x (Tfinal – Tinitial)
Note: This calculation is to change the heat of the system.
Heat capacity is units J/Celcius
The heat capacity of a system is the amount of heat required to
raise the temperature of the system by 1 degree Celcius
Calculate heat of a pure substance
q = m x c x DT
where
q = heat
c = specific heat of pure substance
sum of T = Temp final – Temp initial
The heat capacity of a system is the amount of heat required to
raise the temperature of the system by 1 degree Celcius
Calculate heat of a pure substance
q = m x c x DT
where
q = heat
c = specific heat of pure substance
sum of T = Temp final – Temp initial
retrieved from http://faculty.camosun.ca/larrylee/files/2011/09/thermochemistry.pdf