Experiment 3: Vitamin c assay
Introduction
Vitamin C is generic descriptor for all compounds exhibiting qualitatively the biological activity of ascorbic acid. Ascorbic acid is involved in many phycological functions in living organisms. Its role in the synthesis of collagen in connective tissues is well known. The absence of wound healing and the failure of fractures to repair are classically recognized features of scurvy. These features are attributable to impaired collagen formation due to the lack of vitamin C. Ascorbic acid is a strong reducing agent and readily oxidizes reversibly to dehydroascorbic acid.
ascorbic acid + I2 → 2 I− + dehydroascorbic acid
Ascorbic acid is widely used in the food industry as an additive to foods to improve the taste and to restore the vitamin C loss due to processing and storage. It can prevent oxidation as a preservative or serve as a stabilizer in various food products and beverages. The classical vitamin deficiency disease, scurvy, was demonstrated by Lind to be a dietary deficiency resulting from lack of fresh fruit and vegetables. Result of a clinical experiment indicated that scorbutic patients recovered from disease by drinking lemon juice.
Procedures
Preparing the vitamin C extract
Concept
Determination of vitamin C concentration is based on redox titration method by using iodine. When the iodine is dropped into the Erlenmeyer flask containing ground extract, HCl and starch solution, the ascorbic acid (vitamin C) is oxidized to dehydroascorbic acid while the iodine is reduced to iodide ions.
ascorbic acid + I2 → 2 I− + dehydroascorbic acid.
As long as there is any ascorbic acid present, the iodine would keep reduced to iodide ions. When all the ascorbic acid are all bind to the iodine, the iodine would then bind to the starch molecule and this causes the ground extract solution turns dark blue in colour.
Result
Vitamin C is generic descriptor for all compounds exhibiting qualitatively the biological activity of ascorbic acid. Ascorbic acid is involved in many phycological functions in living organisms. Its role in the synthesis of collagen in connective tissues is well known. The absence of wound healing and the failure of fractures to repair are classically recognized features of scurvy. These features are attributable to impaired collagen formation due to the lack of vitamin C. Ascorbic acid is a strong reducing agent and readily oxidizes reversibly to dehydroascorbic acid.
ascorbic acid + I2 → 2 I− + dehydroascorbic acid
Ascorbic acid is widely used in the food industry as an additive to foods to improve the taste and to restore the vitamin C loss due to processing and storage. It can prevent oxidation as a preservative or serve as a stabilizer in various food products and beverages. The classical vitamin deficiency disease, scurvy, was demonstrated by Lind to be a dietary deficiency resulting from lack of fresh fruit and vegetables. Result of a clinical experiment indicated that scorbutic patients recovered from disease by drinking lemon juice.
Procedures
Preparing the vitamin C extract
- We chose pineapple as our vitamin C source. First, we chopped the pineapple into small pieces and placed it into the blender.
- 100 ml of distilled water was added into the blender.
- The material was blended using the highest speed until it thoroughly ground.
- The ground extract was strained and 30 ml of it was measured into 250ml Erlenmeyer flask.
- The 30ml of ground extract was placed in the 250ml flask.
- Then, 2 drops of 0.1M HCl was dropped into the flask containing ground extract.
- 5 ml of the starch solution was added to the flask.
- A burette was filled with the iodine solution and the initial volume reading was recorded.
- After that, the iodine solution was added in 1ml increments to the flask while swirling the flask.
- The iodine was added until the solution stays blue-black for 15 seconds.
- Lastly, the volume reading on the burette was recorded.
Concept
Determination of vitamin C concentration is based on redox titration method by using iodine. When the iodine is dropped into the Erlenmeyer flask containing ground extract, HCl and starch solution, the ascorbic acid (vitamin C) is oxidized to dehydroascorbic acid while the iodine is reduced to iodide ions.
ascorbic acid + I2 → 2 I− + dehydroascorbic acid.
As long as there is any ascorbic acid present, the iodine would keep reduced to iodide ions. When all the ascorbic acid are all bind to the iodine, the iodine would then bind to the starch molecule and this causes the ground extract solution turns dark blue in colour.
Result
Discussion
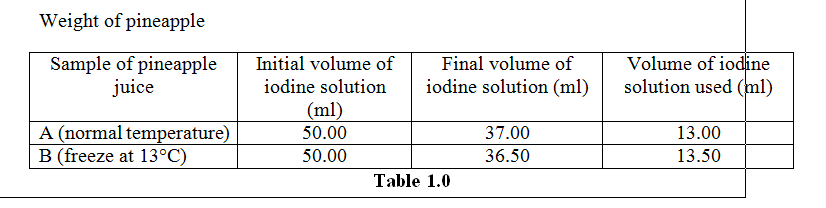
Based on the Table 1.0, the volume of iodine used to oxidize ascorbic acid into hydroascorbic acid is the same as the amount of the vitamin C contained in the pineapple ground extract. The changes of the colour of the ground extract from yellow to dark blue colour indicates that all the ascorbic acid molecules are already bind to the iodine before it binds to starch molecule. At normal temperature, the volume of iodine solution used is 13.00m. At 13°C, the volume of iodine solution used is 12.80mL.
Vitamin C leaching by water and steam is detrimental. It generally preserve during freezing and thawing. Careless blanching and low temperature storage are critical for vitamin C preservation (H.D. Belitz. W.Grosch. P.Schieberle, 2009). As stated in the table 1.0, we can see that the amount of ascorbic acid containing in the sample B is more than A at low temperature. It shows that the lower the temperature the better the concentration of Vitamin C in pineapple juice. Higher temperature does not favour Vitamin C. It is better to maintain or store Vitamin C in a place below the room temperature. Vitamin C is the most unstable vitamin which can easily denature. Vitamin C also denatured on cooling. On freezing, the food containing vitamin C puts a negative effect on it if it is exposed to the air. But during the experiment, we covered the Erlenmeyer flask containing ground extract by using aluminium foil so that the enzymatic action are preserved.
Conclusion
Vitamin C is the most unstable vitamin which can easily denature. This study supports the common perception that vitamin C is preserved as long as it undergoes minimal storage at room temperature.
References
Lester Packer, Fuchs Jurgen. (1997). Vitamin C in health and disease. New York: Marcel Dekker, Inc. 270 Madison Avenue.
H.D. Belitz. W.Grosch. P.Schieberle. (2009). Food Chemistry (4th revised and extended edition). Springer-verleg Berlin Heidilberg
Behind the scene
Based on the Table 1.0, the volume of iodine used to oxidize ascorbic acid into hydroascorbic acid is the same as the amount of the vitamin C contained in the pineapple ground extract. The changes of the colour of the ground extract from yellow to dark blue colour indicates that all the ascorbic acid molecules are already bind to the iodine before it binds to starch molecule. At normal temperature, the volume of iodine solution used is 13.00m. At 13°C, the volume of iodine solution used is 12.80mL.
Vitamin C leaching by water and steam is detrimental. It generally preserve during freezing and thawing. Careless blanching and low temperature storage are critical for vitamin C preservation (H.D. Belitz. W.Grosch. P.Schieberle, 2009). As stated in the table 1.0, we can see that the amount of ascorbic acid containing in the sample B is more than A at low temperature. It shows that the lower the temperature the better the concentration of Vitamin C in pineapple juice. Higher temperature does not favour Vitamin C. It is better to maintain or store Vitamin C in a place below the room temperature. Vitamin C is the most unstable vitamin which can easily denature. Vitamin C also denatured on cooling. On freezing, the food containing vitamin C puts a negative effect on it if it is exposed to the air. But during the experiment, we covered the Erlenmeyer flask containing ground extract by using aluminium foil so that the enzymatic action are preserved.
Conclusion
Vitamin C is the most unstable vitamin which can easily denature. This study supports the common perception that vitamin C is preserved as long as it undergoes minimal storage at room temperature.
References
Lester Packer, Fuchs Jurgen. (1997). Vitamin C in health and disease. New York: Marcel Dekker, Inc. 270 Madison Avenue.
H.D. Belitz. W.Grosch. P.Schieberle. (2009). Food Chemistry (4th revised and extended edition). Springer-verleg Berlin Heidilberg
Behind the scene